|
8/30/2023 0 Comments Keynote 042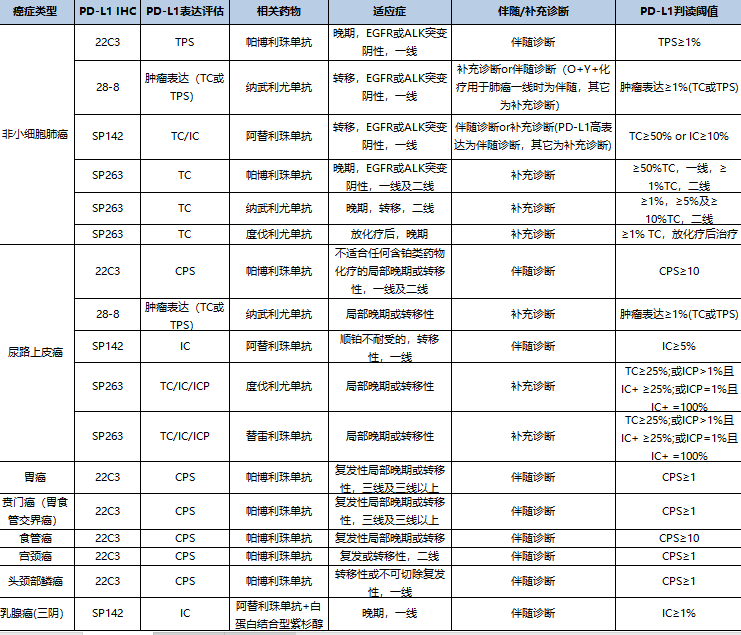
In addition to TPS %, the ECOG-PS has been identified as an additional discriminating parameter because this value reflects the overall clinical status of the patients, indirectly reflecting the immune system’s ability to respond.Ĭurrent clinical decisions for advanced NSCLC in first-line treatment are based on the presence of genetic aberrations, such as sensitizing epidermal growth factor receptor (EGFR) mutations, and translocations of anaplastic lymphoma kinase (ALK). The ECOG scale ranges between 0 (fully active) and 5 (dead). It describes a patients’ level of functioning in terms of self-care, daily activity, and physical ability. The Eastern Cooperative Oncology Group Performance Status (ECOG-PS) is an international scale that standardizes criteria for measuring how cancer impacts the patient’s daily living abilities. 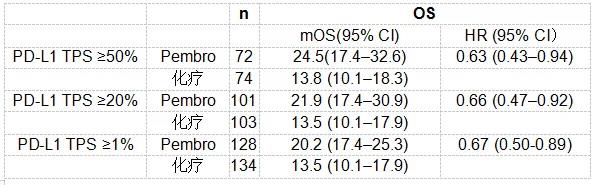
Based on these results, the TPS percentage was identified as a discriminating parameter in this work. ĭata from the phase 1 KEYNOTE-001 and phase 3 KEYNOTE-010 studies indicated that patients with advanced NSCLC and a PD-L1 tumour proportion score (TPS) of 50% or greater are more likely to respond to pembrolizumab than those with a lower TPS. Human and humanized monoclonal antibodies that inhibit the PD-1 receptor, such as nivolumab and pembrolizumab, showed a more favourable tolerance profile and improved results when compared to chemotherapy. The therapeutic blockade of PD-1, expressed on the T cells by the linkage of PD-L1 and PD-L2 expressed by the tumour cells, prevents the downregulation of the T cell effector function, allowing the lymphocytes to mediate tumour cell death. The triggers of inflammatory and immunological reactions are correlated to the mechanism of action and can be exacerbated by genetic predisposition, environmental relation and presence of target antigens in both tumour and involved tissue. In fact, pembrolizumab interferes with the adaptive immune system activation on the PD-1 checkpoint mostly located on the surface of the lymphocytes type T. However, a substantial proportion of patients will not benefit from these treatments, and robust biomarkers are required to help clinicians select patients who are most likely to benefit. In the last decade, immune checkpoint inhibitors (ICIs) targeting programmed death-1 (PD-1) or programmed death-ligand 1 (PD-L1) have proved to be a promising treatment option for patients with advanced/metastatic NSCLC. In particular, NSCLC is the most common type of lung cancer, accounting for 84% of all lung cancer diagnoses, as reported by international cancer statistics ( ). These real-world findings demonstrated the significant correlation between OS and cutaneous toxicities.Īccording to the World Health Organization (WHO), non–small cell lung cancer (NSCLC) is a common and deadly malignancy with over 2.21 million new diagnoses and over 1.8 million associated deaths per year worldwide. The percentage of irAEs detected was comparable to that reported in KEYNOTE-024 and KEYNOTE-042. Patients experiencing any type of toxicity had a significantly longer median OS (20.39 months, 95% CI: 13.08-NA) than those with no toxicities (6.46 months, 95% CI: 1.41-NA, p = 0.006). Immune-Related Adverse Events (irAEs) occurred in 77.5% of patients (30.1% cutaneous, 27.5% gastrointestinal, and 20.4% endocrinological), but no grade 4 or 5 irAEs were identified. The median OS of 13.6 months (95% CI: 11.7-NA) was not influenced by sex and PD-L1, but was significantly associated with ECOG-PS ( p = 0.02). The median number of cycles was 8.5 at a median follow-up of 13 months. The entire cohort had stage IV NSCLC at diagnosis. The cohort’s main characteristics were as follows: median age 73 years, 64.3% were male and 35.7% were female, an ECOG-PS score of 0 ( n = 73) and 1 or 2 ( n = 25), and a PD-L1 > 90% in 29.6% of patients. Clinical data, including PD-L1 expression, Performance Status (ECOG-PS), treatment duration, toxicity, and outcomes were retrieved from local electronic medical records and from the Italian Regulatory Agency Registry. Patients were treated with pembrolizumab (200 mg q3w) as a first-line treatment. This retrospective observational study involved 98 patients with mNSCLC, TPS ≥ 50%, and no EGFR/ALK aberrations. The aim of this study was to reveal the correlation between OS and adverse events in real-world settings after 42 months. Many trials supported pembrolizumab as a first-line monotherapy to significantly improve overall survival (OS) in selected patients with previously untreated metastatic Non–Small Cell Lung Cancer (mNSCLC) and a PD-L1 TPS of ≥50% without EGFR/ALK mutations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
